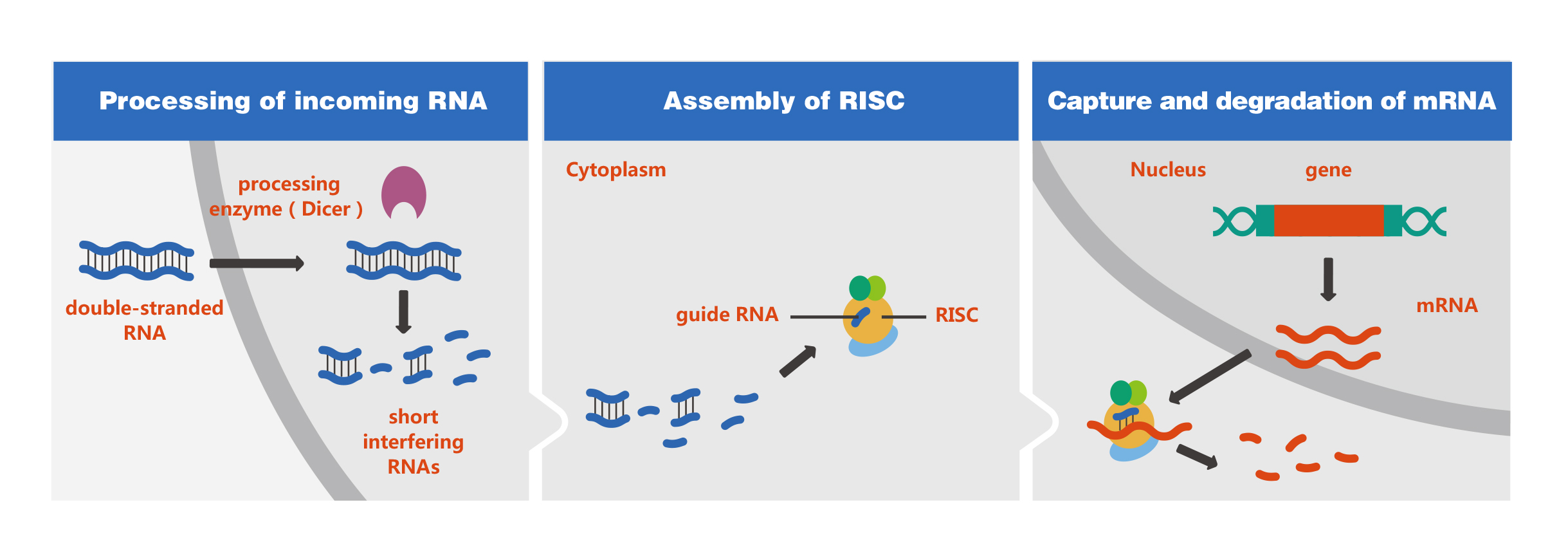
In recent years, RNAi has become an indispensable tool for studying gene function. Its applications have expanded from genomics to medical fields and the development of gene therapies. As such an important molecular biology technology, how RNAi woks? As shown in the figure below: on entry into cells, the exogenous dsRNAs are cleaved by Dicer to yield short interfering RNAs (siRNAs), which subsequently are unwounded and integrated into RNA-induced silencing complex (RISC), a multimeric protein complex with endonuclease activity. After integration into the RISC, siRNAs base-pair to the target mRNA and cleave it, thereby inhibiting protein expression.
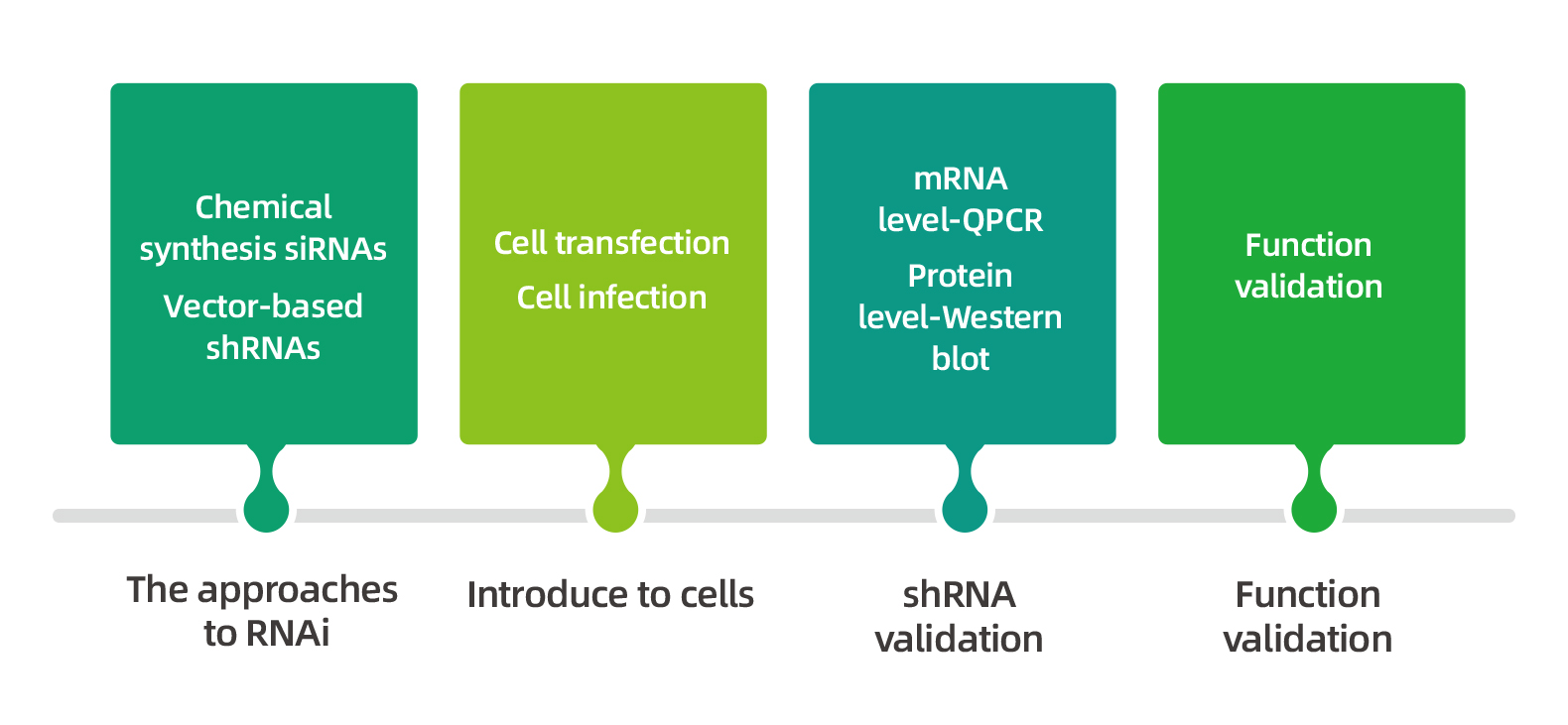
According on the RNAi mechanism, a typical RNAi experiment can be roughly divided into four steps, see the figure below:
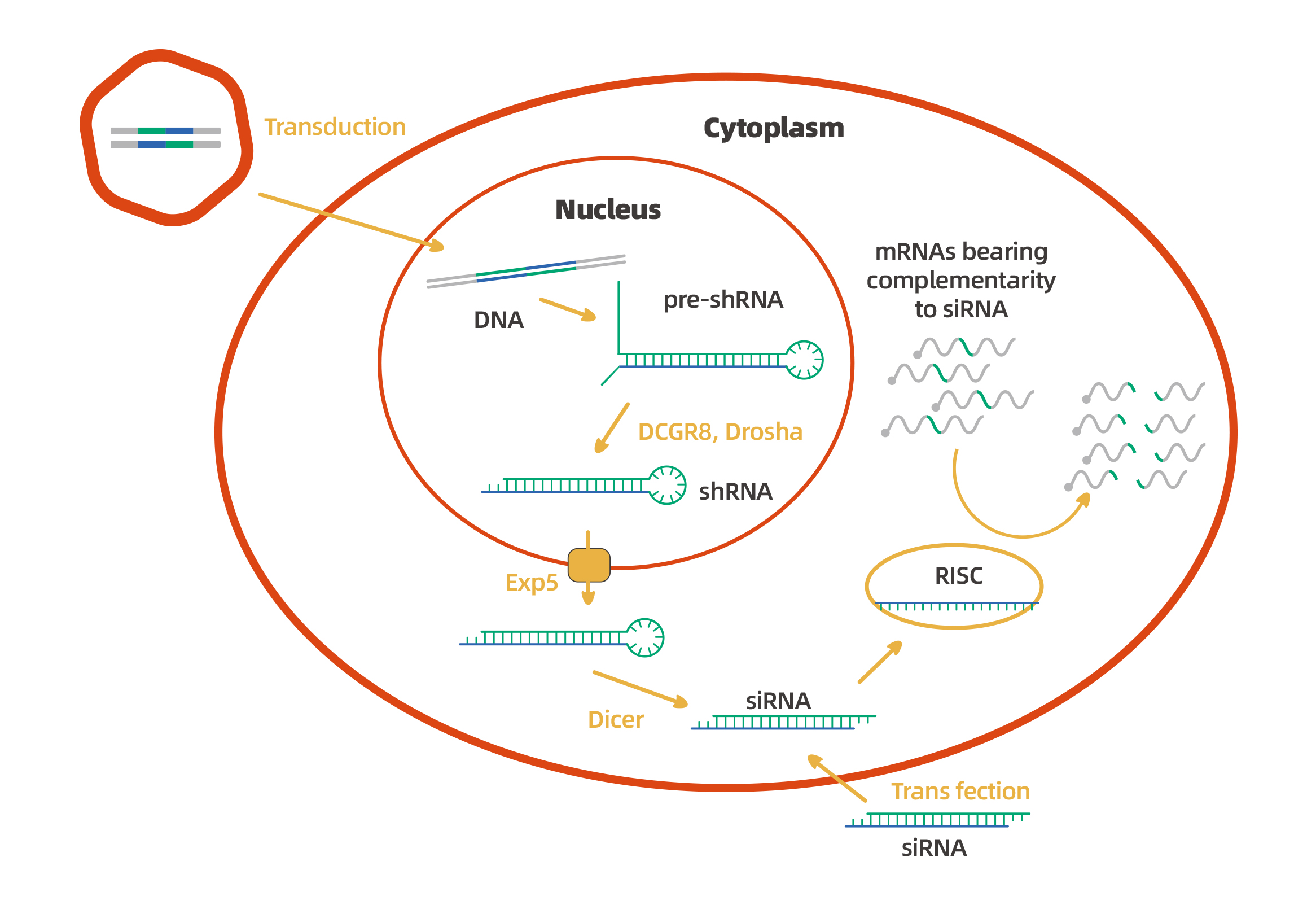
Besides the chemically synthesized siRNAs, WZ Biosciences can provide full spectrum of RNAi services(vector-based shRNAs), including shRNA construction, cell introduction and inhibition effect verification. The siRNAs are the effector molecule of RNAi. No matter how they are produced, they will eventually bind with RISC, active RISC then binds to target mRNA and cleave it, thereby silencing target gene expression.
*The shRNA, called a short hairpin RNA, is an artificial RNA molecule that can be cloned into an shRNA expression vector and processed into siRNAs.
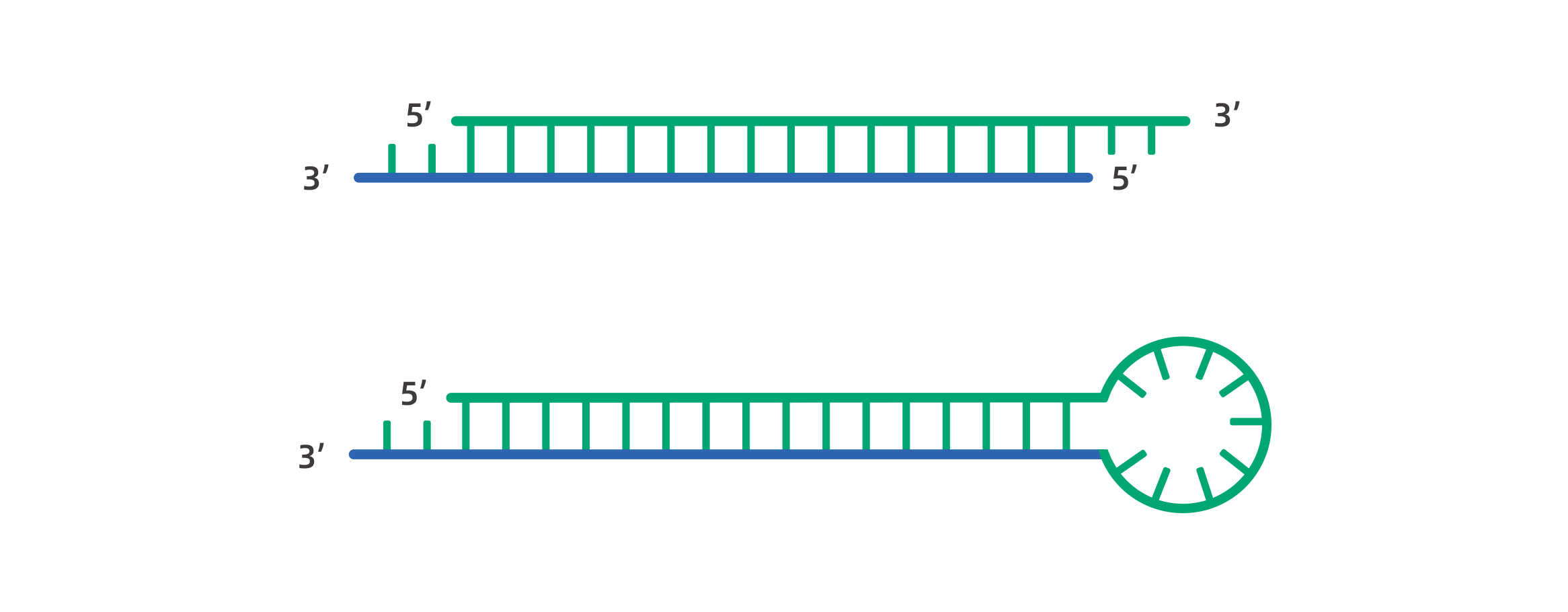
The siRNA, also known as small interfering RNA, is a class of double-stranded RNA molecules of 21-23 nucleotides (nt) with 2-3 nucleotides 3’ overhang and phosphorylated 5’ends & hydroxylated 3’ends. The dsRNAs are cleaved by Dicer(RNase III-related endonuclease), thereby mediating RNA interference. The structure of siRNA and shRNA is shown in figure below.
Although both siRNAs and shRNAs can be used for gene silencing or gene knockdown, there are differences in their mechanisms of action. See the figure below:
Comparison of siRNA and shRNA:
|
Feature
|
shRNA
|
siRNA
|
|
Stability
|
High
|
poor, rapidly degraded, short half-life
|
|
Persistence
|
Long-term target KD (several weeks or even longer)
|
Short-term target KD(aboat 1 week)
|
|
Off-target effects
|
Low
|
High
|
|
Delivery to the cell
|
Via plasmid transfection;
Via viral vector Infection, a means to reach hard-to-transfect cell types for KD
|
Via lipids transfection, some cell types are not transfected efficiently
|
|
Expression
|
Driven by polymerase type II and type III promoters; inducible expression system can be used; co-expression with reporter gene can be used to visualize cell introduction efficiency
|
Chemical sythesis
|
|
KD detection time
|
Depending on the half-life of the target protein
|
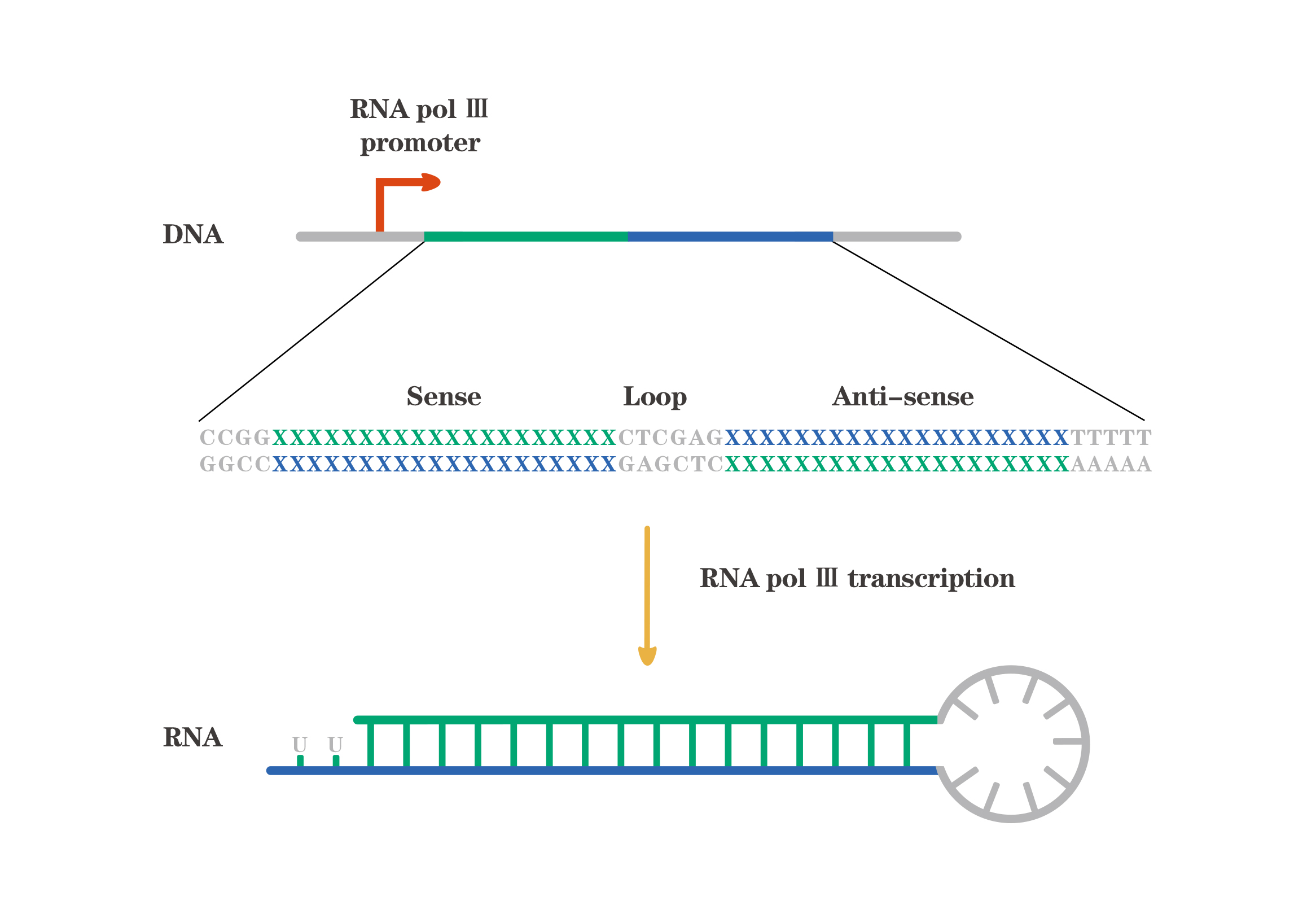
Vector-based shRNAs expression is most commonly driven by the Pol III promoters. The two most commonly used Pol III promoters are U6 and H1. The RNA pol III promoters usually lack tissue-specific, and mainly used to express small RNAs terminated by a stretch of four to six consecutive T. The structure of Vector-based shRNAs is shown below:
Regarding vector-based shRNAs, in order to meet a huge variety of your research requirements, WZ Biosciencess can offer a variety of shRNA plasmid construction services:
1 shRNA construct
You can send us your validated shRNA sequences, then we can clone your shRNA sequences into our shRNA vectors for you.
The set of shRNA constructs
According to your target gene, WZ Biosciences’ scientists design 3 or 4 shRNAs per gene and provide a set of 3-4 plasmids for you, to ensure at least 70% knockdown of every target gene.
Multi-in-1 shRNA construct
WZ Biosciences's proprietary technology can put multiple shRNAs in one expression vector. You can either send us your validated shRNA sequences or provide us with target gene information.
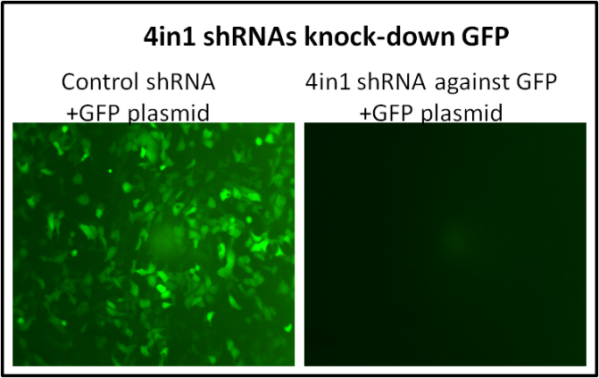
Take 4-in-1 shRNA construct as an example: 4 shRNAs are cloned into one shRNA vector. Our 4-in-1-shRNA vectors can not only be transfected into cells, but also packaged into virus for viral delivery.
|
|
|
Knock down overexpression GFP
|
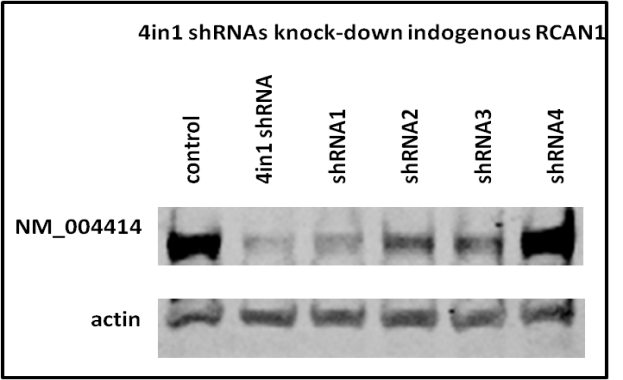
Knock down endogenous RCAN1
|
Advantages
1) Knock down one gene with 4 different shRNAs in one vector
Four shRNA sequences targeted to different sites of mRNA are cloned into one shRNA vector, enhancing the probability of suppression of target gene.
2) Knock down up to 4 different genes using one single plasmid vector
The validated shRNAs against 2-4 target genes are cloned into one shRNA vector, achieving simultaneous down-regulation of multiple target genes.
3) More effective, and far more rapid, compared to the set of shRNA
Researchers don't need to perform effective shRNA screen, which greatly reduce their workload.
4) Complete shRNA knockdown vector system
Multiple 4-in-1 shRNA viral vectors are available, including adenovirus vectors, lentivirus vectors and adeno-associated virus vectors.
Disadvantages
Because 4 shRNAs are simultaneously expressed, we are not sure which one is the best for down-regulation of the target gene.
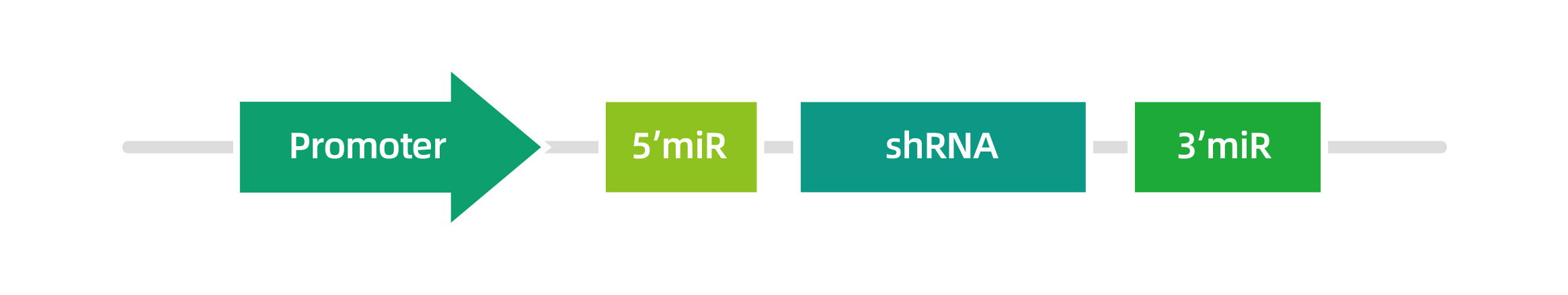
miR30-based shRNA construct, achieving tissue-specific down-regulation of the target gene.
The mechanism of shRNA action is similar to the microRNA maturation pathway. Therefore, shRNAs can also be designed to be transcribed by the Pol II promoters, including ubiquitous promoters (such as CMV, CAG and EF1a, etc.) and tissue-specific promoters (such as hsyn, TBG and cTnT, etc.). The Pol II promoters can utilized for the transcription of the long genes, and their termination is mediated through the poly(A)-dependent pathway. WZ Biosciences can clone your shRNA sequences into our miR-30-based shRNA vectors which allows for stable expression of shRNAs from customer-specified promoters. Structure of mir30-based-shRNAs is shown below:
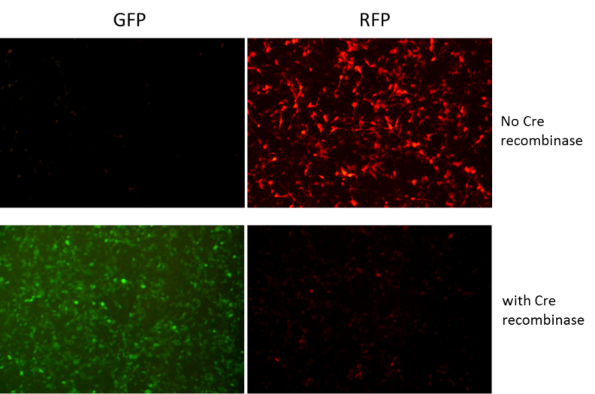
Inducible shRNA Construct, including miR30-based Cre-inducible, miR30-based Flp-inducible and miR30-based tetracycline-inducible RNAi system.
Take Cre-ON shRNA as an example, the investigators can use the FLEx (flip-excision) system to achieve Cre-mediated conditional shRNA expression. FLEx is a very powerful approach for controlling gene expression. In FLEX system, the DNA sequence is flanked by two pairs of loxP and lox2272 sites in opposing orientations and the Cre will invert the sequence between the sites permanently. Our FLEx system is improved that two pairs of lox sites in opposing orientations are located on both sides of promoter, so the presence or absence of Cre determines the orientation of the promoter, and different fluorescence can be used as an indicator of shRNA expression.

|
《Transfection Reagent Instructions》
|
|
1. Which promoters can drive WZ Biosciences’s vector-based shRNAs?
In WZ Biosciences, multiple promoters can be used to drive shRNA expression in cells, including Pol III promoters, U6 and H1, and Pol II promoters, ubiquitous promoters (such as CMV, CAG and EF1a, etc.) and tissue-specific promoters (such as hsyn, TBG and cTnT, etc.).
2. Can human U6 and H1 work in rat, mouse or other mammalian cells?
In general, human H6 and H1 should all work in human, mouse, rat and other mammalian cells.
3. Do you guarantee knockdown efficiency?
For the set of shRNA constructs, we guarantee that at least one shRNA construct will generate ≥70% knockdown of every target gene(determined by qPCR), under the condition of transfection efficiency of >80% in target cells. If none of them can reach this knockdown efficiency, we will reconstruct new shRNA plasmids for free, which requires an additional turnaround time. If none of the re-constructed shRNA vectors still can’t achieve this knockdown efficiency, it may be associated with the target gene, we only charge you one time.
4. For every target gene, what you suggest for shRNA cloning services?
For every target gene, we suggest that you can send us you validated shRNA sequence or choose our the set of shRNA constructs.
5. How can I monitor transfection efficiency?
Our shRNA vectors contains GFP or RFP reporters which allow stimatanous transfection monitoring.
6. Do you construct shRNA vectors with other shRNA destination vectors purchased from other company?
Yes, we do. You just need to send us your desired shRNA vectors.
7. How long is the turnaround time for Vector-based shRNA?
Vector-based shRNA often need take 2-3 weeks.
8.What are the advantages of viral vectors used for gene delivery?
Viral-based shRNAs are ideal for hard-to-difficult cells, such as primary cells, fibroblast cells and dendritic cells (DC).
|
1.
|
Cell Death and Disease. (IF=6.304). Tian, et al. (2020). MYC-regulated pseudogene HMGA1P6 promotes ovarian cancer malignancy via augmenting the oncogenic HMGA1/2.
|
|
|
2.
|
INTERNATIONAL JOURNAL OF ONCOLOGY. (IF=3.899). Cui, et al. (2019). TGF-β-induced long non-coding RNA MIR155HG promotes the progression and EMT of laryngeal squamous cell carcinoma by regulating the miR-155-5p/SOX10 axis.
|
|
|
3.
|
Cerebral Cortex. (IF=5.043). Lu, et al.(2017). Neuritin Enhances Synaptic Transmission in Medial Prefrontal Cortex in Mice by Increasing CaV3.3 SurfaceExpression.
|
|
|
4.
|
Journal of Cellular and Molecular Medicine. (IF=4.486). Wang, et al. (2020). Inhibition of PFKFB3 suppresses osteoclastogenesis and prevents ovariectomy-induced bone loss.
|
|
|
5.
|
EBioMedicine. (IF=5.736. Li, et al. (2019). Inhibition of AZIN2-sv induces neovascularization and improves prognosis after myocardial infarction by blocking ubiquitin-dependent talin1 degradation and activating the Akt pathway.
|
|
|
6.
|
Bone. (IF=4.147). Zhao, et al. (2018). YAP1 is essential for osteoclastogenesis through a TEADs-dependent mechanism.
|
|
|
7.
|
Cardiovascular Research. (IF=8.168. Li, et al. (2018). Loss of AZIN2 splice variant facilitates endogenous cardiac regeneration.
|
|
|
8.
|
Cell Discovery. (IF=6.255). Liao, et al. (2019). USP10 modulates the SKP2/Bcr-Abl axis via stabilizing SKP2 in chronic myeloid leukemia.
|
|
|
9.
|
Oncogene. (IF=7.971). Li, et al. (2019). MicroRNA-150 Modulates Adipogenic Differentiation of Adipose-Derived Stem Cells by Targeting Notch3.
|
|
|
10.
|
Oncogene. (IF=7.971). Liao, et al. (2018). Growth arrest and apoptosis induction in androgen receptor-positive human breast cancer cells by inhibition of USP14-mediated androgen receptor deubiquitination.
|
|
|
11.
|
J Mol Cell Cardiol. (IF= 4.133). Li Z, et al. (2017).TCONS_00075467 modulates atrial electrical remodeling by sponging miR-328 to regulate CACNA1C.
|
|
|
12.
|
Behavioural Neurology. (IF=2.093). Qu, et.al. (2018). MST1 Suppression Reduces Early Brain Injury by Inhibiting the NF-κB/MMP-9 Pathway after Subarachnoid Hemorrhage in Mice.
|
|